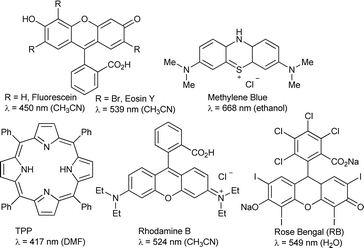
Visible-light initiated aerobic oxidations: a critical review - Green Chemistry (RSC Publishing) DOI:10.1039/C8GC02382D

The Development of Visible‐Light Photoredox Catalysis in Flow - Garlets - 2014 - Israel Journal of Chemistry - Wiley Online Library
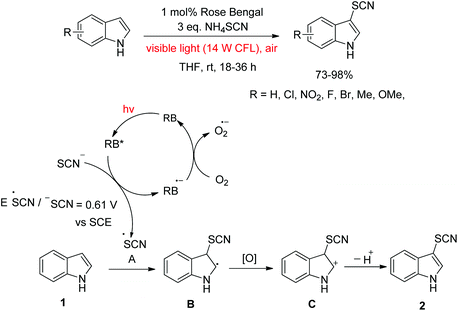
Dehydrogenative coupling reactions catalysed by Rose Bengal using visible light irradiation - Green Chemistry (RSC Publishing) DOI:10.1039/C1GC15489C

Modeling of the 5 '-Deiodination of Thyroxine by Iodothyronine Deiodinase: Chemical Corroboration of a Selenenyl Iodide Intermediate

Recent advances in the synthesis of oxaspirolactones and their application in the total synthesis of related natural products - Organic & Biomolecular Chemistry (RSC Publishing)

Conference Issue: A cluster of articles from speakers attending the 5th EuCheMS Chemistry Congress, Istanbul: Chemistry – A European Journal: Vol 20, No 35













