Thiol−Ene Reaction: Synthetic Aspects and Mechanistic Studies of an Anti‐Markovnikov‐Selective Hydrothiolation of Olefins - Sinha - 2019 - Asian Journal of Organic Chemistry - Wiley Online Library
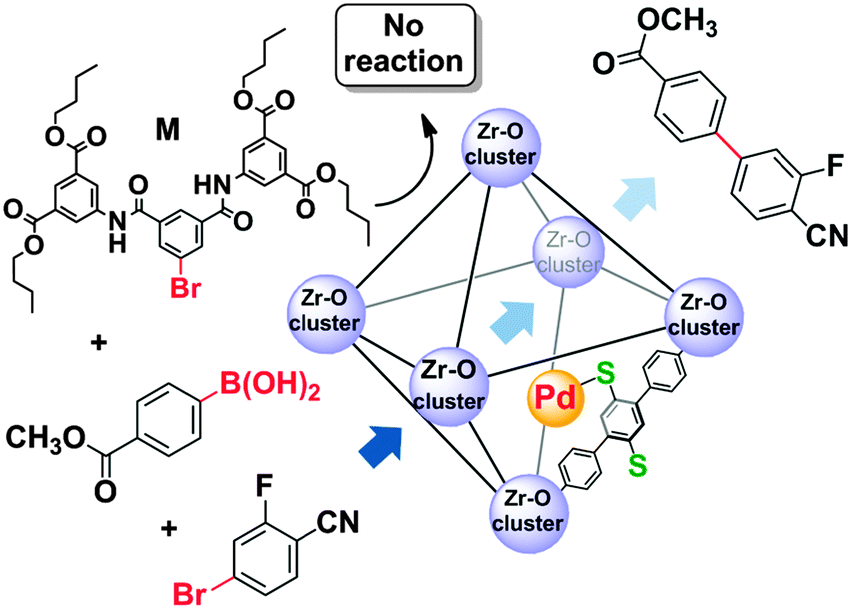
Tackling poison and leach: catalysis by dangling thiol–palladium functions within a porous metal–organic solid - Chemical Communications (RSC Publishing)

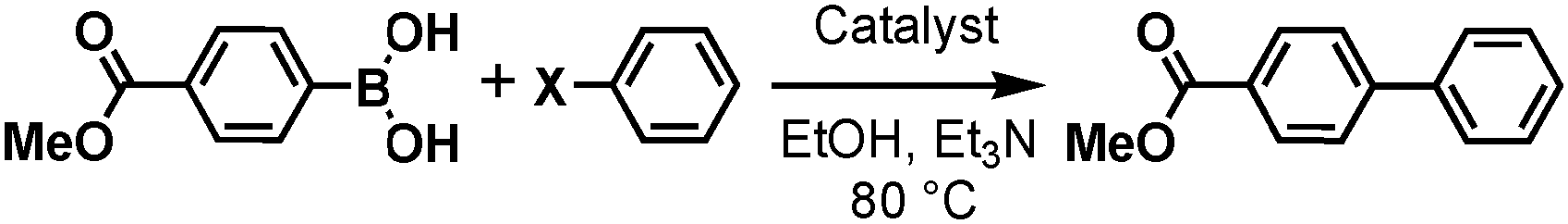
Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds
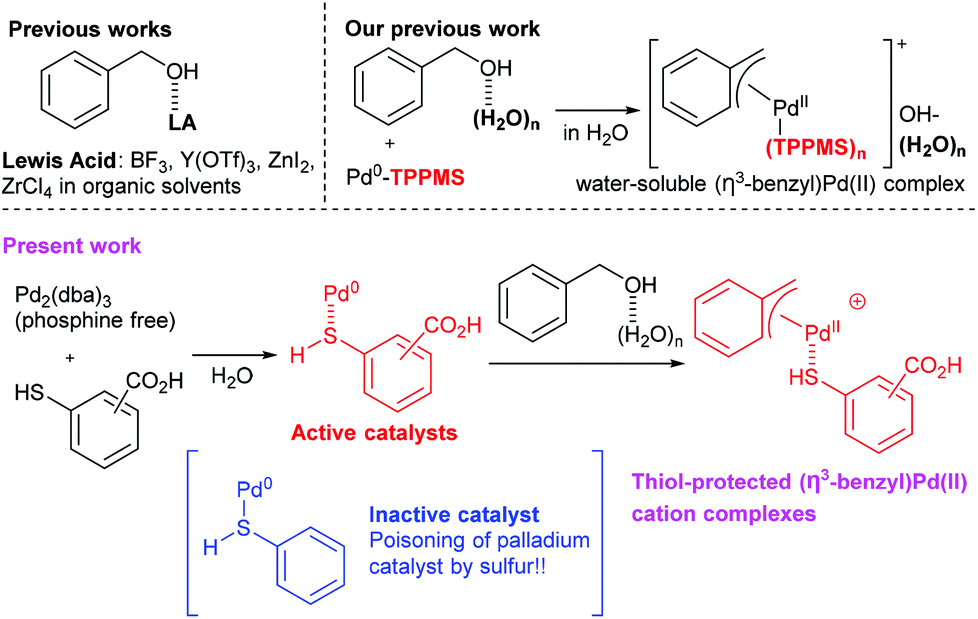
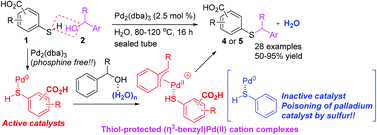
Mercaptobenzoic acid-palladium(0) complexes as active catalysts for S-benzylation with benzylic alcohols via (η3-benzyl)palladium(ii) cations in water - Organic & Biomolecular Chemistry (RSC Publishing)

Palladium‐Catalyzed C–S Bond Formation of Stable Enamines with Arene/Alkanethiols: Highly Regioselective Synthesis of β‐Amino Sulfides - Jiang - 2016 - European Journal of Organic Chemistry - Wiley Online Library
Tackling poison and leach: catalysis by dangling thiol–palladium functions within a porous metal–organic solid - Chemical Communications (RSC Publishing)
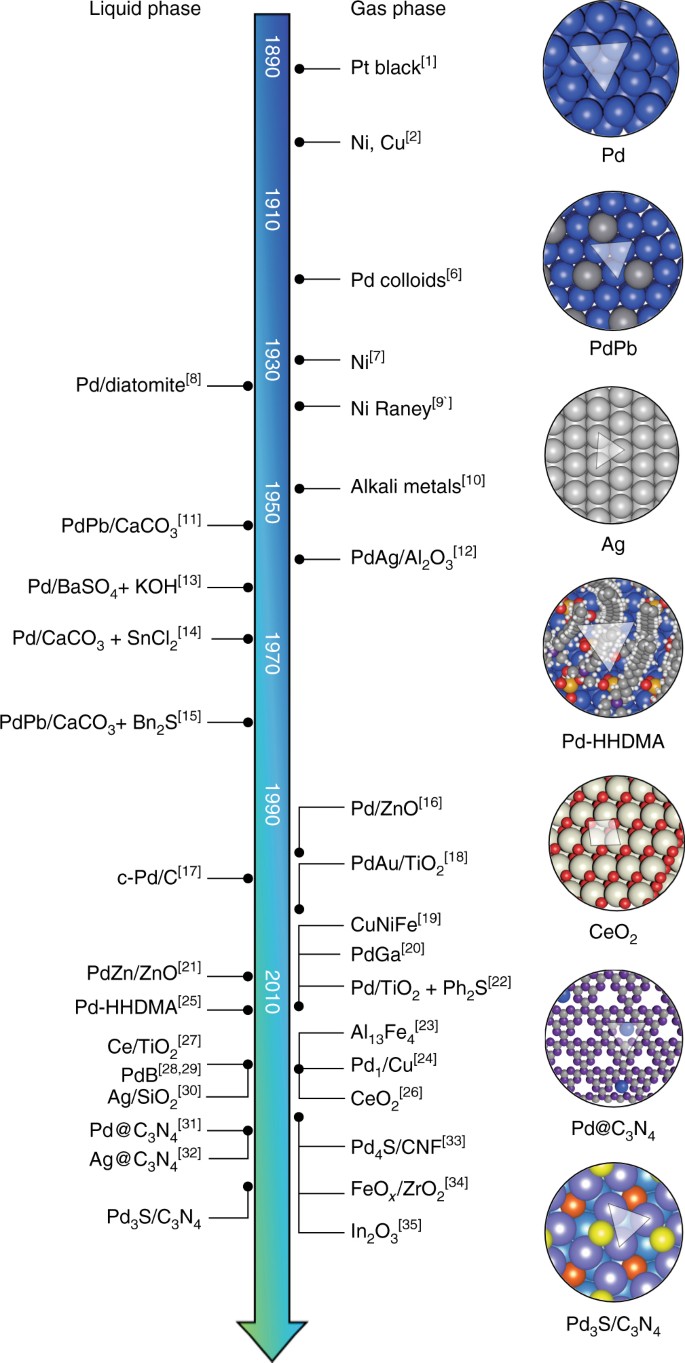
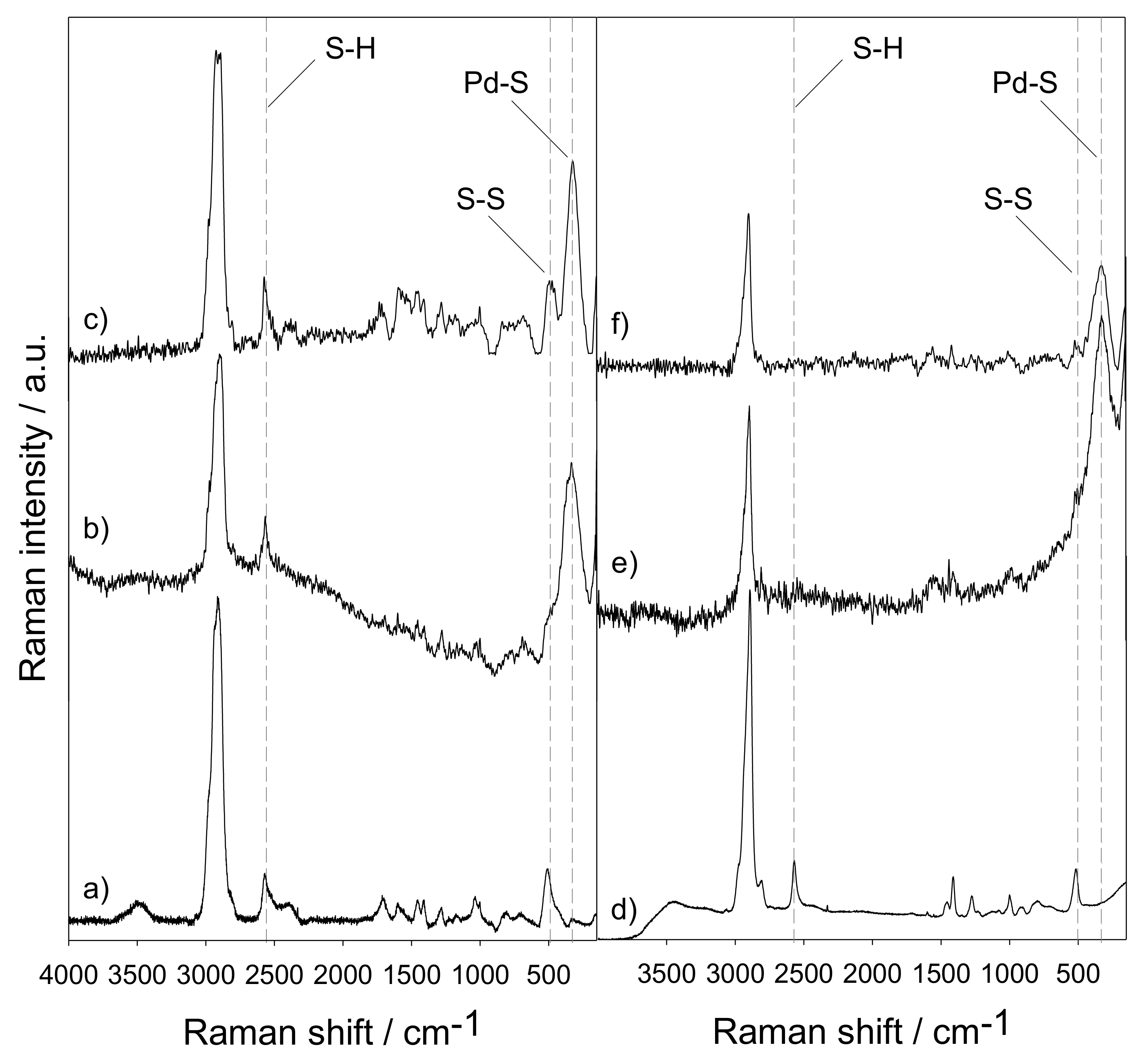
Selective ensembles in supported palladium sulfide nanoparticles for alkyne semi-hydrogenation | Nature Communications

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Thiol Treatment Creates Selective Palladium Catalysts for Semihydrogenation of Internal Alkynes - ScienceDirect

Thiol Treatment Creates Selective Palladium Catalysts for Semihydrogenation of Internal Alkynes - ScienceDirect

Pd/BIPHEPHOS is an Efficient Catalyst for the Pd‐Catalyzed S‐Allylation of Thiols with High n‐Selectivity - Schlatzer - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library

Optimising surface d charge of AuPd nanoalloy catalysts for enhanced catalytic activity | Nature Communications

Palladium-catalyzed carbon-sulfur or carbon-phosphorus bond metathesis by reversible arylation | Science
Materials | Free Full-Text | Thiol-Functionalized Ethylene Periodic Mesoporous Organosilica as an Efficient Scavenger for Palladium: Confirming the Homogeneous Character of the Suzuki Reaction | HTML