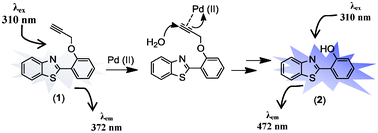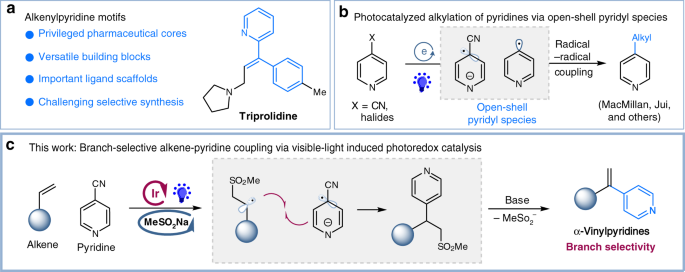
Photoredox-catalyzed branch-selective pyridylation of alkenes for the expedient synthesis of Triprolidine | Nature Communications

Recent Developments in Pd0‐Catalyzed Alkene‐Carboheterofunctionalization Reactions - Garlets - 2017 - Asian Journal of Organic Chemistry - Wiley Online Library
![Palladium-catalyzed synthesis and fluorescence study of 2,3-diaryl-5-ethynylbenzo[e]indoles - ScienceDirect Palladium-catalyzed synthesis and fluorescence study of 2,3-diaryl-5-ethynylbenzo[e]indoles - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0040402017303848-fx1.jpg)
Palladium-catalyzed synthesis and fluorescence study of 2,3-diaryl-5-ethynylbenzo[e]indoles - ScienceDirect
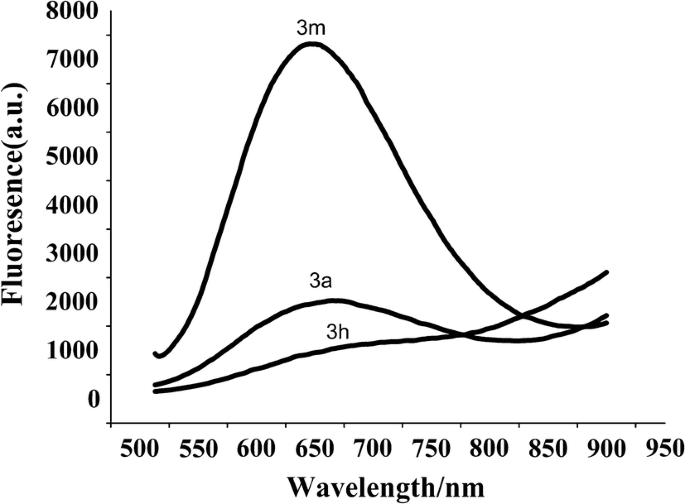
Synthesis of (1 E ,3 E )-1,4-diarylbuta-1,3-dienes promoted by μ-OMs palladium–dimer complex | SpringerLink

Phosphaannulation of Aryl‐ and Benzylphosphonic Acids with Unactivated Alkenes via Palladium‐Catalyzed CH Activation/Oxidative Cyclization Reaction - Jeon - 2015 - Advanced Synthesis & Catalysis - Wiley Online Library

Scheme 1 Synthesis of TTZ-1 and schematic of its fluorescent response... | Download Scientific Diagram
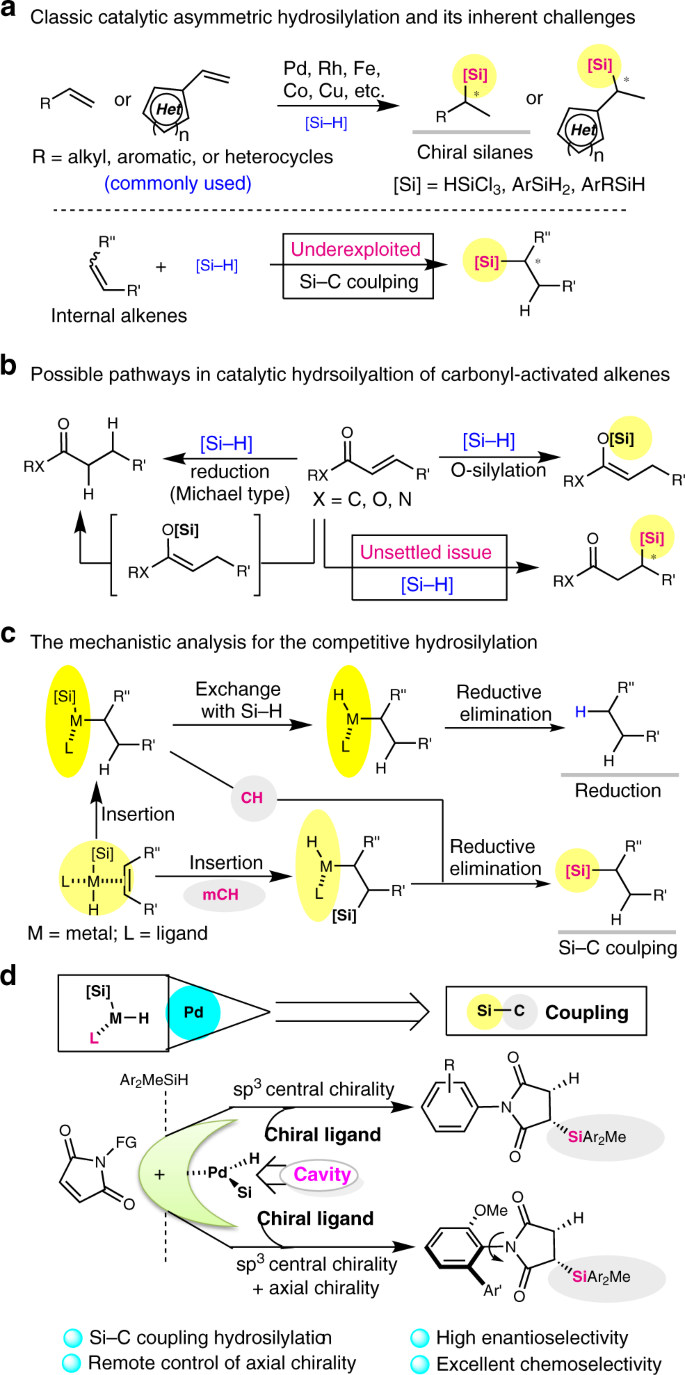
Stereospecific Si-C coupling and remote control of axial chirality by enantioselective palladium-catalyzed hydrosilylation of maleimides | Nature Communications
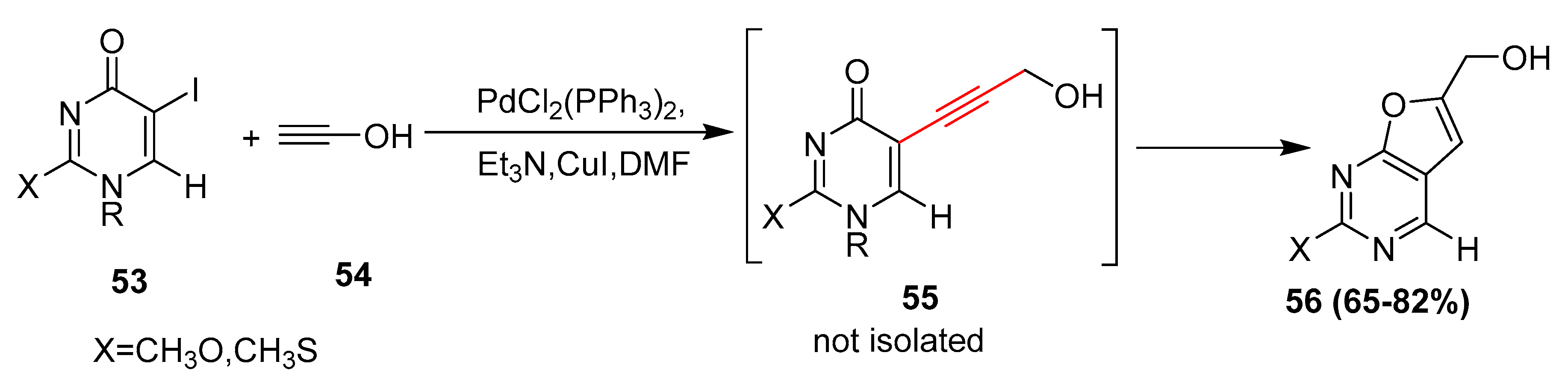
Catalysts | Free Full-Text | Palladium and Copper Catalyzed Sonogashira cross Coupling an Excellent Methodology for C-C Bond Formation over 17 Years: A Review | HTML

Palladium‐Catalyzed Synthesis of Benzophenanthrosilines by C−H/C−H Coupling through 1,4‐Palladium Migration/Alkene Stereoisomerization - Tsuda - - Angewandte Chemie - Wiley Online Library

Expeditious Synthesis of Tetrasubstituted Helical Alkenes by a Cascade of Palladium‐Catalyzed CH Activations - Liu - 2012 - Angewandte Chemie International Edition - Wiley Online Library

N-alkylation of 2-pyridone derivatives via palladium(II)-catalyzed directed alkene hydroamination - ScienceDirect
![Synthesis of Substituted Benzo[b]thiophenes via Sequential One-Pot, Copper-Catalyzed Intermolecular C-S Bond Formation and Palladium-Catalyzed Intramolecular Arene-Alkene Coupling of Bis(het)aryl/alkyl-1,3-monothiodiketones and o-Bromoiodoarenes ... Synthesis of Substituted Benzo[b]thiophenes via Sequential One-Pot, Copper-Catalyzed Intermolecular C-S Bond Formation and Palladium-Catalyzed Intramolecular Arene-Alkene Coupling of Bis(het)aryl/alkyl-1,3-monothiodiketones and o-Bromoiodoarenes ...](https://d3i71xaburhd42.cloudfront.net/95cff7feec2fffaff4d5ae744c1d00c294992095/2-Table1-1.png)
Synthesis of Substituted Benzo[b]thiophenes via Sequential One-Pot, Copper-Catalyzed Intermolecular C-S Bond Formation and Palladium-Catalyzed Intramolecular Arene-Alkene Coupling of Bis(het)aryl/alkyl-1,3-monothiodiketones and o-Bromoiodoarenes ...

Highly selective electrochemical hydrogenation of alkynes: Rapid construction of mechanochromic materials | Science Advances
![Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/us/category/images/00074-img02.png)
Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation
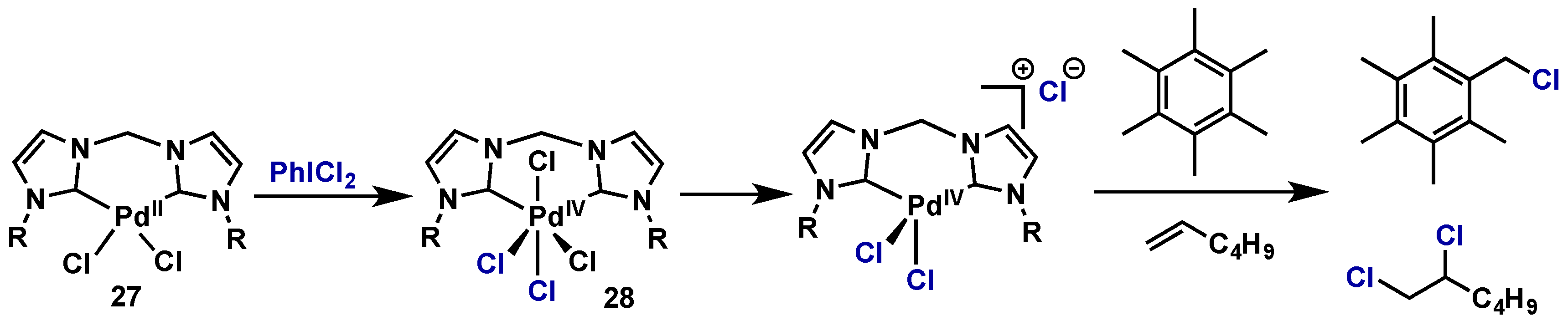
Molecules | Free Full-Text | Hypervalent Iodine Reagents in High Valent Transition Metal Chemistry | HTML

Mechanism and Origin of MAD-Induced Ni/N-Heterocyclic Carbene-Catalyzed Regio- and Enantioselective C-H Cyclization of Pyridines with Alkenes. - Chem. Eur. J. - X-MOL
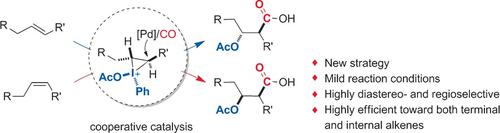
A Cooperative Strategy for the Highly Selective Intermolecular Oxycarbonylation Reaction of Alkenes using a Palladium Catalyst - Angew. Chem. Int. Ed. - X-MOL

PDF) Pd/P( t -Bu) 3 : A Mild Catalyst for Selective Reduction of Alkenes under Transfer-Hydrogenation Conditions
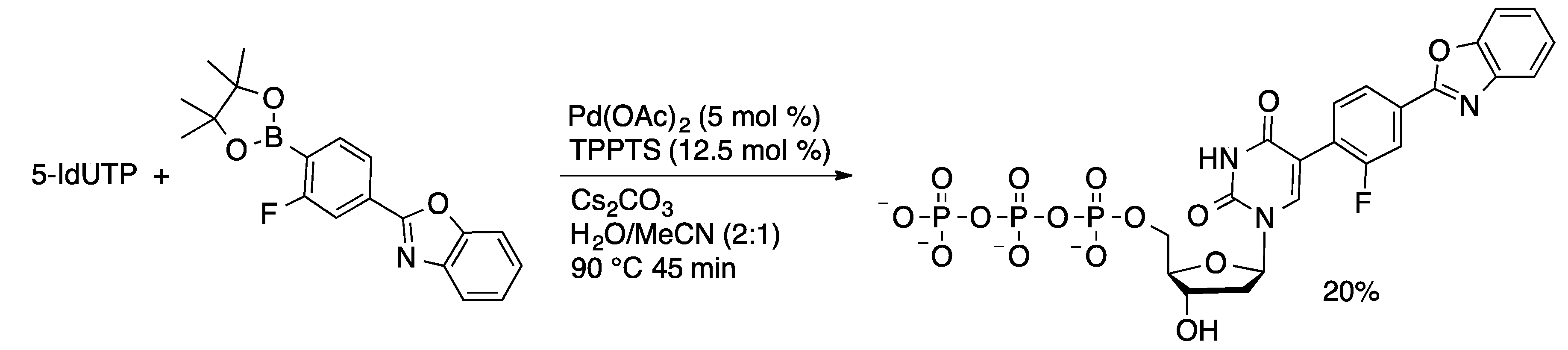
Molecules | Free Full-Text | Palladium-Catalyzed Modification of Unprotected Nucleosides, Nucleotides, and Oligonucleotides | HTML

1,2,3‐Trisubstituted Indanes by Highly Diastereoselective Palladium‐Catalyzed Oxyarylation of Indenes with Arylboronic Acids and Nitroxides - Kirchberg - 2010 - Angewandte Chemie International Edition - Wiley Online Library

Scheme 1 Reactions used to demonstrate the catalytic farming concept.... | Download Scientific Diagram

Molecules | Free Full-Text | Asymmetric Palladium-Catalysed Intramolecular Wacker-Type Cyclisations of Unsaturated Alcohols and Amino Alcohols | HTML