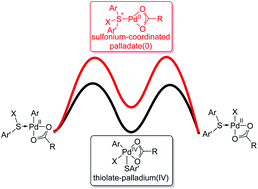
Mechanism of Palladium-Catalyzed Alkylation of Aryl Halides with Alkyl Halides through C–H Activation: A Computational Study - Organometallics - X-MOL

Pd Palladium Ball Powder Pure 99.98% Periodic Table of Rare earth Precious Metal Elements Research Study Education Collection|table periodic|table tabletable periodic table - AliExpress

First–Principles Study Including Zero Point Energy on Hydrogen in Palladium for Hydrogen Membranes Applications | Scientific.Net

In situ LTEM study of the dissolution of Pd nanocubes using a liquid... | Download Scientific Diagram

Highly efficient one pot palladium-catalyzed synthesis of 3,5-bis (arylated) pyridines: Comparative experimental and DFT studies - ScienceDirect
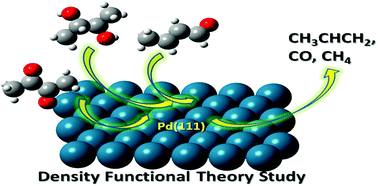
First-principles study on the gas-phase decomposition of bio-oil oxygenated compounds over the palladium catalyst surface - Physical Chemistry Chemical Physics (RSC Publishing)

Palladium-catalyzed carbonylative synthesis and theoretical study of elongated tubular cavitands - ScienceDirect
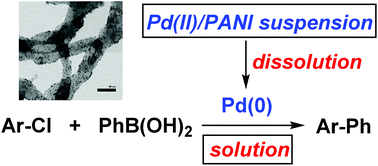
A mechanistic study of cross-coupling reactions catalyzed by palladium nanoparticles supported on polyaniline nanofibers - Inorganic Chemistry Frontiers (RSC Publishing)
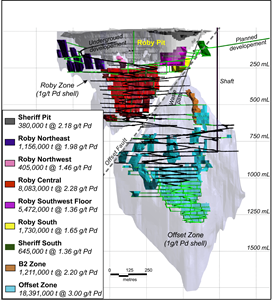
North American Palladium Completes a New Feasibility Study Featuring a Major Expansion of Underground Reserves Toronto Stock Exchange:PDL

Mechanistic insight into the electrocatalytic hydrodechlorination reaction on palladium by a facet effect study - ScienceDirect
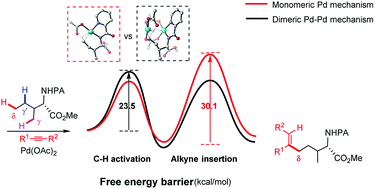
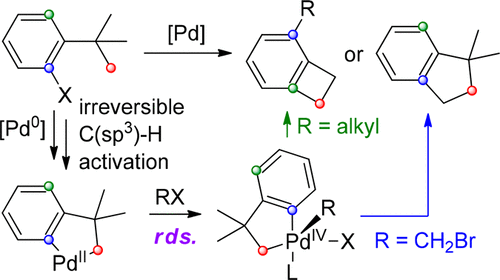
Computational study on palladium-catalyzed alkenylation of remote δ-C(sp3)–H bonds with alkynes: a new understanding of mechanistic insight and origins of site-selectivity - RSC Advances (RSC Publishing)

Map showing study sites tested for platinum (Pt) and palladium (Pd):... | Download Scientific Diagram
Determination of Platinum, Palladium, and Lead in Biological Samples by Atomic Absorption Spectrophotometry
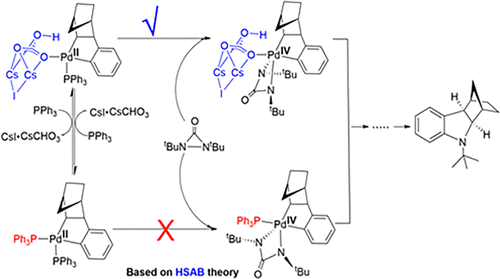
DFT Study on the Mechanism of Palladium(0)-Catalyzed Reaction of Aryl Iodides, Norbornene, and Di-tert-butyldiaziridinone - Organometallics - X-MOL
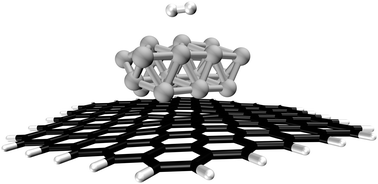
A quantum chemical study of hydrogen adsorption on carbon-supported palladium clusters - Physical Chemistry Chemical Physics (RSC Publishing)

Novartis Reports Results of QMF149 in P-III PALLADIUM Study for Patients with Uncontrolled Asthma | PharmaShots

DFT study on the mechanism of palladium(II)-catalyzed reaction of allyl-substituted 3,4-dienoate, alkyne and carbon monoxide - ScienceDirect
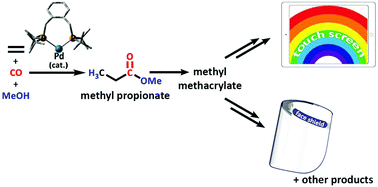
Palladium-catalysed methoxycarbonylation of ethene with bidentate diphosphine ligands: a density functional theory study - Physical Chemistry Chemical Physics (RSC Publishing)
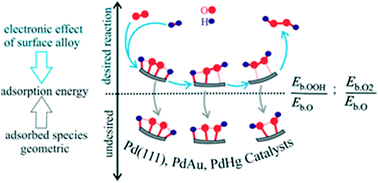
Descriptor study by density functional theory analysis for the direct synthesis of hydrogen peroxide using palladium–gold and palladium–mercury alloy catalysts - Molecular Systems Design & Engineering (RSC Publishing)

Investigation of DNA binding and bioactivities of thioether containing Schiff base Copper(II), Cobalt(II) and Palladium(II) complexes: Synthesis, characterization, spectrochemical study, viscosity measurement - ScienceDirect
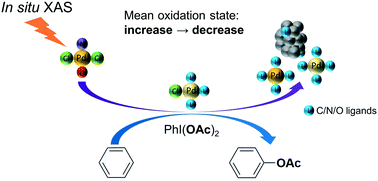
In situ XAS study of the local structure and oxidation state evolution of palladium in a reduced graphene oxide supported Pd(ii) carbene complex during an undirected C–H acetoxylation reaction - Catalysis Science

Intermolecular metallophilic interactions in palladium(II) chalcogenolate compounds – An experimental and theoretical study - ScienceDirect
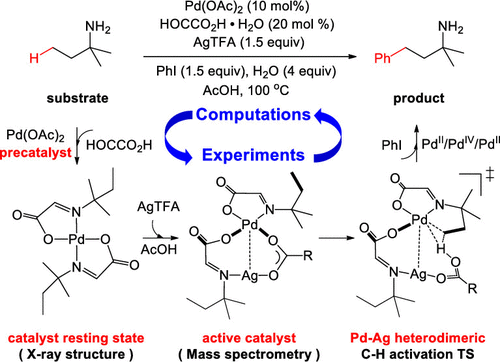
Mechanism of the Palladium-Catalyzed C(sp3)–H Arylation of Aliphatic Amines: Unraveling the Crucial Role of Silver(I) Additives - ACS Catal. - X-MOL

Computational approach of palladium (II) complex ions with binuclear diamine ligands thermo-physical, chemical, and biological properties: a dft study
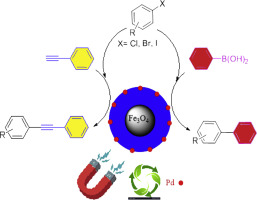
Efficient heterogenization of palladium by citric acid on the magnetite nanoparticles surface (Nano-Fe3O4@CA-Pd), and its catalytic application in C-C coupling reactions - J. Organomet. Chem. - X-MOL