![Palladium-catalysed [π2a+π2s] cycloadditions of α-bromoalkyl ketenes to cyclopentadiene - ScienceDirect Palladium-catalysed [π2a+π2s] cycloadditions of α-bromoalkyl ketenes to cyclopentadiene - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0040403900004780-sc2.gif)
Palladium-catalysed [π2a+π2s] cycloadditions of α-bromoalkyl ketenes to cyclopentadiene - ScienceDirect

Chiral Lewis acid catalysts in diels-Alder cycloadditions: mechanistic aspects and synthetic applications of recent systems
![Palladium-catalysed [π2a+π2s] cycloadditions of α-bromoalkyl ketenes to cyclopentadiene - ScienceDirect Palladium-catalysed [π2a+π2s] cycloadditions of α-bromoalkyl ketenes to cyclopentadiene - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0040403900004780-fx1.gif)
Palladium-catalysed [π2a+π2s] cycloadditions of α-bromoalkyl ketenes to cyclopentadiene - ScienceDirect
![Enantioselective Synthesis of 2,2‐Disubstituted Tetrahydrofurans: Palladium‐Catalyzed [3+2] Cycloadditions of Trimethylenemethane with Ketones - Trost - 2013 - Angewandte Chemie International Edition - Wiley Online Library Enantioselective Synthesis of 2,2‐Disubstituted Tetrahydrofurans: Palladium‐Catalyzed [3+2] Cycloadditions of Trimethylenemethane with Ketones - Trost - 2013 - Angewandte Chemie International Edition - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/73b53f64-7b6e-4d08-9570-7e998e7e6c06/mcontent.jpg)
Enantioselective Synthesis of 2,2‐Disubstituted Tetrahydrofurans: Palladium‐Catalyzed [3+2] Cycloadditions of Trimethylenemethane with Ketones - Trost - 2013 - Angewandte Chemie International Edition - Wiley Online Library
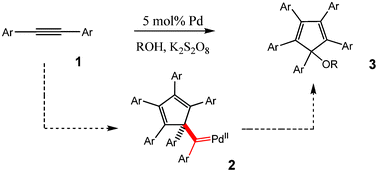
Synthesis of cyclopentadienyl alkyl ethers via Pd-catalyzed cyclotrimerization of diarylacetylenes - RSC Advances (RSC Publishing)
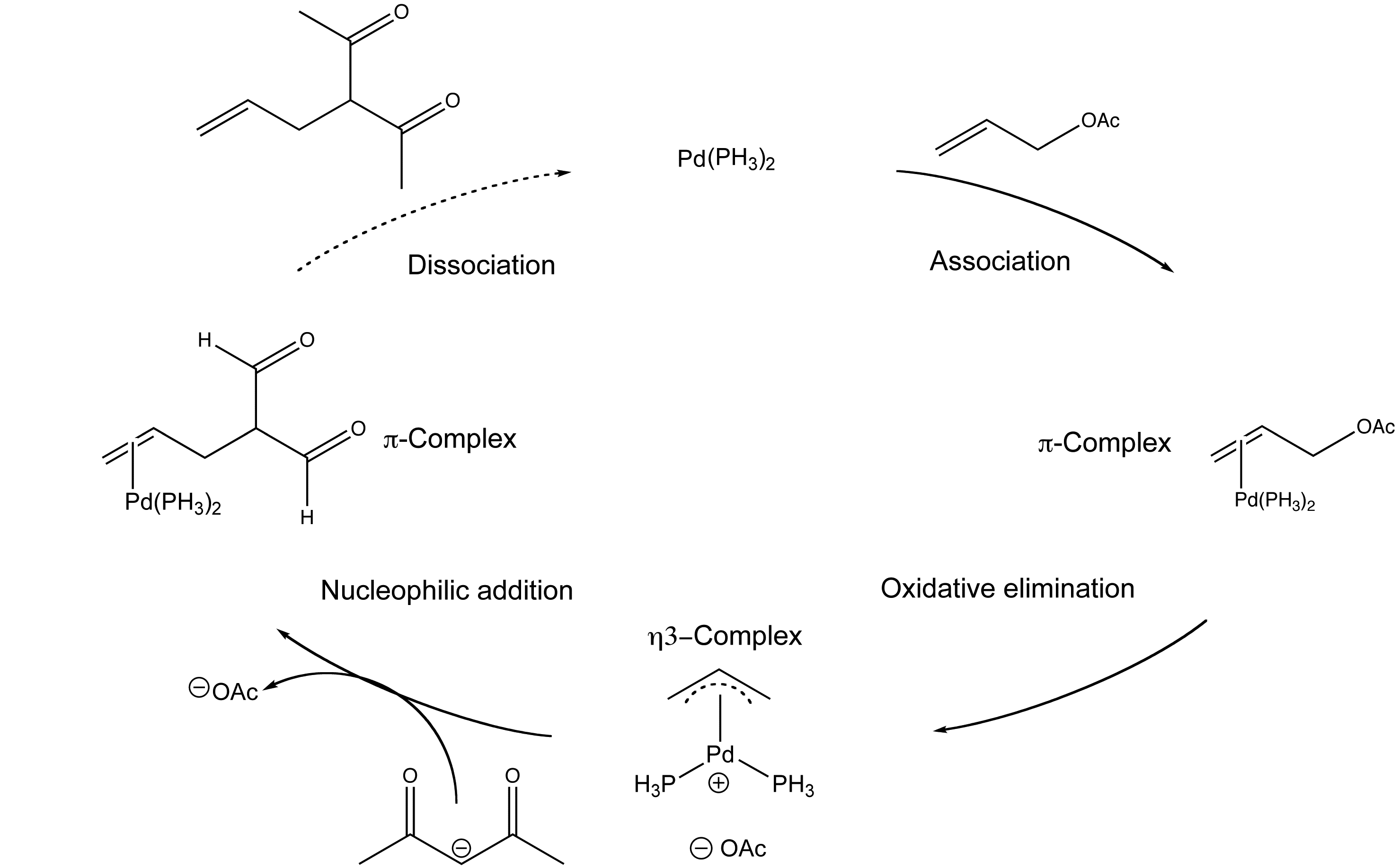
Organopalladium Chemistry - Palladium-catalysed nucleophilic allylic substitution of functionalised compounds

Influence of palladium(II) complexes on the cycloaddition of α-bromoalkyl ketenes to cyclopentadiene - ScienceDirect

Influence of palladium(II) complexes on the cycloaddition of α-bromoalkyl ketenes to cyclopentadiene - ScienceDirect

Two‐Step, Stereoselective Hydrazidoarylation of 1,3‐Cyclopentadiene - Yao - 2002 - Angewandte Chemie International Edition - Wiley Online Library
UvA-DARE (Digital Academic Repository) Transition Metal Catalysis Using Core-Functionalized Carbosilane Dendrimers. van Oosterom

Facile Access to Cyclopentadienes via Catalytic Intramolecular Palladium-Ene Reaction of 2,4-Pentadienyl Acetates - Synlett - X-MOL
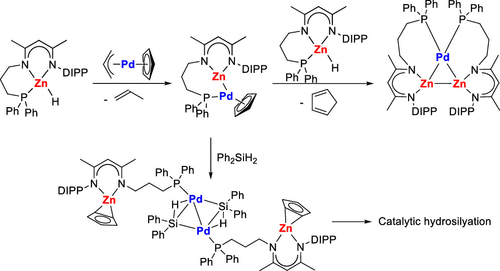
Formation of Zn–Zn and Zn–Pd Bonded Complexes by Reactions of Terminal Zinc Hydrides with Pd(II) Species - Inorg. Chem. - X-MOL
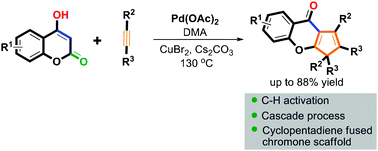
Palladium-catalyzed cascade reactions of coumarins with alkynes: synthesis of highly substituted cyclopentadiene fused chromones - Chemical Communications (RSC Publishing)
![Palladium-catalysed [π2a+π2s] cycloadditions of α-bromoalkyl ketenes to cyclopentadiene - ScienceDirect Palladium-catalysed [π2a+π2s] cycloadditions of α-bromoalkyl ketenes to cyclopentadiene - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0040403900004780-fx2.gif)
Palladium-catalysed [π2a+π2s] cycloadditions of α-bromoalkyl ketenes to cyclopentadiene - ScienceDirect

Influence of palladium(II) complexes on the cycloaddition of α-bromoalkyl ketenes to cyclopentadiene - ScienceDirect
![Palladium-catalysed [π2a+π2s] cycloadditions of α-bromoalkyl ketenes to cyclopentadiene - ScienceDirect Palladium-catalysed [π2a+π2s] cycloadditions of α-bromoalkyl ketenes to cyclopentadiene - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0040403900004780-sc1.gif)